
For several years now, hydrogen has been identified by many experts as the fuel of the future. And there is no denying that this resource has many advantages. It exists in abundance on Earth, its combustion releases only water and it is easily extracted from the many substances that contain hydrogen atoms, such as water or all hydrocarbons. But despite these many qualities, the market for hydrogen vehicles remains small, with a very limited offering and fewer than fifty cars sold in France last year.
How can we explain this paradox? Can hydrogen really become the fuel of a decarbonised and sustainable mobility? This paper attempts to take stock of this emerging technology and to examine how it can be a powerful solution for all types of mobility.
Hydrogen vehicles: how do they work?
Before going into detail about how hydrogen vehicles work, let’s look at the nature of this gas and the ways in which it can be produced. The hydrogen atom is the simplest chemical element, the most abundant on Earth and the oldest in the universe, having first appeared 13 billion years ago. Hydrogen exists naturally on Earth, held in gaseous form in the “geological traps” formed by the different layers of the continental or oceanic subsoil. The discovery of significant and exploitable deposits is relatively recent, but exploitation is still in its infancy, unlike natural gas and other fossil fuels (oil, coal, etc.). On the other hand, hydrogen is very common in its combined state in many substances, in particular with oxygen, with which it forms water (H2O), and with carbon, with which it forms all hydrocarbons.
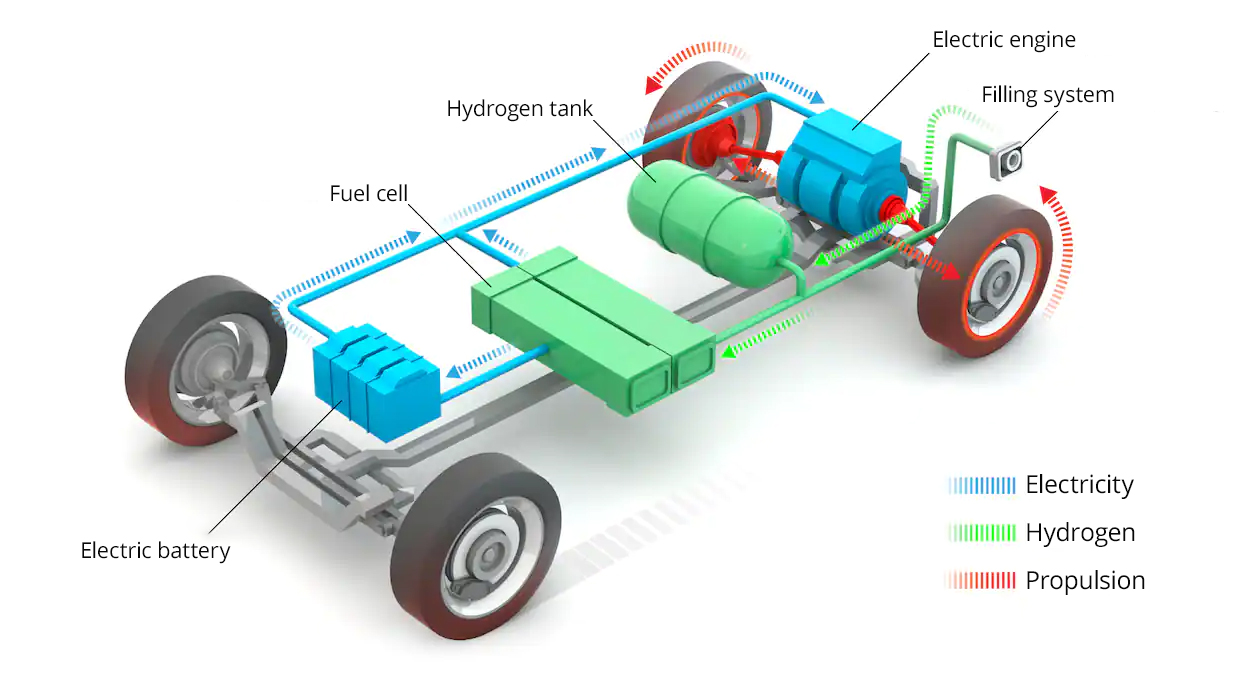
The calorific value of hydrogen is the highest of all existing fuels. At equivalent mass, it is two to three times higher than oil or natural gas. Unlike hydrocarbons, the combustion of hydrogen does not produce any harmful emissions, in particular climate-damaging gases such as CO2, but also health-damaging gases such as carbon oxides. A hydrogen vehicle produces only water vapour and oxygen, at least if it is powered by a fuel cell (FC). Indeed, there are two ways to use hydrogen to power an engine. The first is to use it instead of petrol by burning it in an internal combustion engine, as is the case with NASA’s rockets. These vehicles are known as HICEVs (Hydrogen Internal Combustion Engine Vehicles), a technique developed as early as 1806 by the Swiss inventor Isaac de Rivaz but strangely forgotten about for two centuries until the Hydrogen 7 model, of which BMW sold about a hundred in the 2000s. The second is the use of a hydrogen fuel cell that produces the electricity needed to run the electric motor. The common abbreviation for this type of fuel cell vehicle is FCEV (Fuel Cell Electric Vehicle).
The important difference between FCEVs and other electric or hybrid vehicles is that the hydrogen FCEV itself generates the electricity it needs instead of using energy from an integrated battery. In the FC, the hydrogen in the tanks reacts with the oxygen supplied by the ambient air in a chemical reaction called reverse electrolysis, which produces only electrical energy, heat and water, which is discharged as steam through the exhaust.
How does an hydrogen fuel cell work?
The electrical energy generated in this way is used to power the vehicle’s electric motor and to charge a battery, which is used for charging during acceleration, for example. This battery is significantly smaller and lighter than an EV battery because it is continuously powered by the fuel cell. In addition, FCEVs recover braking energy, which converts the vehicle’s kinetic energy into electrical energy stored in the buffer battery.
Since hydrogen does not exist (or only to a limited extent) in its natural form on Earth, it must be produced or rather extracted from the raw materials that contain it by dissociating it from the other atoms with which it is combined. Dihydrogen (H2) can thus be produced by several chemical or thermal processes. The most widely used, especially in industry, is steam reforming of natural gas, which involves reacting methane and steam at high temperature (700 °C) to split the carbon atoms and produce a synthesis gas containing dihydrogen and carbon dioxide.
This process accounts by far for the majority of current dihydrogen production due to its low cost (€2 per kg of H2) and simplicity. However, it is very polluting and generates a lot of greenhouse gases. The same is true of coal gasification, where a solid compound (usually charcoal) is burned to produce a hydrogen-rich gas. The only non-polluting process for producing hydrogen is water electrolysis, a reaction that uses an electric current to break down water molecules and produce hydrogen and oxygen. The advantage of the latter process is that it has a very low carbon footprint if a decarbonised source of electricity is used (renewable, nuclear, biomass, biomethane).
Depending on the raw material and energy source used, hydrogen will be considered fossil, low carbon or renewable. The major disadvantage of the electrolysis process is its price, which is five times higher than that of steam reforming. As a result, 95% of the hydrogen produced in the world today comes from fossil resources. However, producing one kilo of H2 with this technique produces 10 kg of CO2, i.e. 60 kg of CO2 emitted for a full tank of fuel that allows you to travel 600 km. This is unacceptable if we are to make our vehicles clean. One way of improving this could be the capture and storage of the CO2 emitted by the steam reforming of natural gas. This can be buried underground or processed into new products such as plastic or building materials. This low-carbon hydrogen and its extraction technique could prove to be an acceptable transitional solution, until electrolysis and renewable energy production technologies are sufficient.
Indeed, even if water electrolysis processes can be considered ready for industrial deployment, current research points to significant efficiency gains. The CEA (French Atomic Energy Commission) has discovered a high-temperature electrolysis process (700 °C) that could improve the efficiency of electrolysers to 90%. In Australia, researchers at Curtin University are working on the catalyst used in electrolysis. Instead of the rare and expensive platinum currently used in electrodes, they have developed a material made of iron and sulfur, which is inexpensive and would perform better than the most advanced and expensive catalysts on the market. Clearly, this new material could well be the key to mass-produced, environmentally friendly hydrogen in the future. A sign of the times that the public authorities believe in the future of this clean hydrogen: the thirty or so countries that have adopted a “Hydrogen Plan” in recent years. France is not to be outdone with its May 2018 “Hydrogen Deployment Plan for the Energy Transition”, which aims to green 10% of H2 in 2023 and 20-40% in 2028. In total, France is expected to invest nearly €10 billion between now and 2030. For her part, the European Commissioner for Transport wants there to be “a station at least every 150 km” by 2030, with the aim of putting 60,000 hydrogen-powered trucks on the road.
Pros and cons of hydrogen vehicles
The prospect of the end of sales of combustion engine vehicles by 2035 in Europe is a powerful incentive for governments and manufacturers to accelerate the development of alternative, mainly electric, powertrains. As we have seen, the principle of the hydrogen engine has several apparent advantages over the Battery Engine Vehicle (BEV).
From a driving point of view, an FCEV offers the same driving comfort as a BEV: instantaneous thrust, linear flow, jerk-free pick-up and first-class comfort with no engine noise. Another advantage is the charging time. Depending on the charging station and battery capacity, and despite continuous progress, it takes from 30 minutes to several hours to recharge the battery of a BEV, compared to less than 5 minutes for the FCEV. As far as range is concerned, the hydrogen engine has a clear advantage, as a full tank gives a range of around 700 km. The Toyota Mirai, one of the two models currently available on the French market, has even covered more than 1,000 km in one go in economy driving. It is not surprising to find this model in the Parisian fleet of Hype taxis, with several hundred vehicles currently in use and giving full satisfaction.

A Hype taxi, in the Paris region
If the hydrogen vehicle offers so many advantages for the user and for the environment, why is it that its market remains small and that manufacturers are reluctant to develop new models? The major shortcoming of these vehicles at the moment is the limited refuelling possibilities. Currently, there are very few hydrogen stations. There are barely a hundred of them in France, while the network of charging points for electric vehicles already numbers several tens of thousands. In fact, a hydrogen pump costs almost a million euros, about 20 times more than a 350 kW ultra-fast electric charging station. “When it comes to the hydrogen engine, we have a chicken-and-egg problem,” explains Axel Rücker, Program Manager Hydrogen Fuel Cell at BMW. “As long as the network of hydrogen stations is so small, low customer demand will not allow for cost-effective mass production of FCEVs. And as long as very few hydrogen vehicles are seen on the road, operators will be in no hurry to expand their pump network.” BMW is preparing to release its iX5 Hydrogen SUV in 2023, but only in small volumes. To further the development of the infrastructure, BMW has joined forces with hydrogen producers and service station operators in the Clean Energy Partnership initiative. The company has set its sights on 400 stations by 2025, which would allow some 60,000 hydrogen-powered cars to drive on German roads.
Another factor that explains the low demand for hydrogen vehicles is price. The few mid- to high-range FCEV models available on the market, namely the Toyota Mirai and the Hyundai Nexo, cost almost €70,000, twice as much as a comparable electric or hybrid car. This high price is due to the lack of industrialisation of production and the cost of the platinum used in the fuel cell, even though the quantity required has fallen in recent years. Another reason for the high price is the size of the FCEV. The hydrogen tank(s) take up a lot of space, which makes the vehicle unavoidably large. Hence the absence of small FCEV models, unlike pure electric cars. In addition to the acquisition cost, running costs also play an important role in the acceptance of a particular powertrain technology. Currently, a kilo of H2 costs almost €10 if it comes from a renewable or low-carbon energy source, but without taxes or profitability. Thus, the cost per kilometre of a hydrogen vehicle are currently almost twice as high as those of an electric vehicle charged at home. However, Axel Rücker believes that these costs will even out: “If the demand for hydrogen increases, the price per kilo should fall to around €5 by 2030.” By integrating recent progress in the efficiency of electrolysers, we are now able to lower the price of wind-generated hydrogen stored on site to around €6 per kilo. In any case, “green” hydrogen – hydrogen from a decarbonised energy source – even if it is two to three times cheaper, will probably be more expensive for motorists than electricity stored in a battery.
Can hydrogen become the fuel of ecological and sustainable mobility?
The ecological ideal would be a vehicle that runs only on renewable energy and does not produce harmful emissions. Hydrogen vehicles do not emit any pollutants into the atmosphere other than water vapour. The impact on the climate depends on how the hydrogen is produced. If the electricity used in the electrolysis process comes from renewable energy sources, the climate balance will be neutral. If, on the other hand, the power comes from fossil fuels, the balance will be negative. Everything depends on the electricity mix used, and from this point of view, the hydrogen vehicle is no different from battery electric vehicles.
The use of hydrogen as an energy carrier involves several steps: H2 production, storage, transport and distribution, and conversion of the stored energy into electricity. One disadvantage of hydrogen production is the losses during electrolysis. In industrial electrolysis, one litre of water and 5 kWh of electricity are needed to produce 1,000 litres of dihydrogen. Given the very low density of H2, this gas must be compressed to 700 bar before the fuel cell can produce electricity. This fuel cell has an efficiency of 60%. In fact, the overall energy efficiency “of the electricity used at the wheel” is barely 30%, less than half that of a battery electric vehicle.
For transport and storage, the trend is towards liquid hydrogen, which is easier to transport and store. However, this is more complex and requires more energy than petrol or diesel, at least for the moment. Hydrogen sceptics often argue that to obtain the energy equivalent of one tanker of petrol, 22 identical tankers of H2 in 200 bar cylinders or 3 tankers of liquefied hydrogen are needed. The problem is the same if we imagine a pipeline network that requires the gas to be compressed before it is injected into the network, which entails a much greater expense for hydrogen than for methane.
However, unlike fossil fuels, hydrogen can be produced wherever power and water are available, theoretically right next to service stations. In fact, from an economic and ecological point of view, the only sustainable solution is the production of hydrogen in situ. The Uno-X network stations in Norway and Denmark sell a kilo of renewable hydrogen at around €10 per kilo, but without taxes and with zero profitability. That is more expensive than the most expensive electricity in the world...
While the future of hydrogen as an environmentally friendly fuel for the average person’s car is still uncertain, its use as an efficient storage medium for renewable energy has immense potential. Indeed, the problem with solar or wind power is that it is not necessarily available when you need it and vice versa. However, progress in electrolysers, and in particular the development by the CEA-Liten (Innovation Laboratory for New Energy and Nanomaterial Technologies) of a high-temperature system with an efficiency of up to 90%, would make it possible to reduce the cost of renewable hydrogen to between €6 and €8 per kilo. A valid calculation considering that the initial energy is surplus, i.e. free. In fact, hydrogen appears to be an effective means of storing intermittent electricity, such as wind or solar power that would otherwise be lost, on a massive scale and for long periods of time.
The young Nantes-based company Lhyfe is going to test a floating wind turbine connected to an electrolyser on a floating platform 20 km off the coast of Loire-Atlantique. This is a world first and a real challenge to deal with the problems of seawater desalination, corrosion and platform operation in an isolated environment in difficult marine conditions. Offshore hydrogen could represent 3 GW by 2030, the equivalent of three nuclear reactors.

The real challenge of hydrogen is thus the local production of energy. Storing electricity in the form of hydrogen therefore facilitates this local production, regardless of the means of production. The issue of H2 transport therefore becomes secondary to that of local energy production and storage.?
This is a major issue in the energy and environmental transition to which hydrogen can provide a solution. As renewable electricity generation becomes more important, and the energy system evolves from centralised demand-driven generation to decentralised generation that is increasingly dependent on renewables, the need to store large amounts of electricity reliably, economically and flexibly becomes unavoidable. Hydrogen can thus offer electricity and gas networks the flexibility they need, by storing electricity generated by intermittent renewable energies in the form of liquid or gaseous H2. The hydrogen produced in this way can even be used to inject it into the natural gas distribution network in a certain proportion to form a new fuel gas. Hydrogen thus makes it possible to bridge the gap between the electricity and gas networks (“Power-to-gas”) by optimising the electricity system as a whole.
Finally, the safety of hydrogen vehicles must be addressed, given the highly flammable nature of this gas. The tanks are made of particularly resistant composite materials. They undergo draconian tests before being approved, sometimes including machine gun fire. Their behaviour is observed in more extensive crash and fire tests than with conventional combustion engine vehicles. As a result, hydrogen fuel cell cars are potentially much less dangerous than equivalent petrol models. In the event of a leak alert, the hydrogen detectors in the vehicle activate the solenoid valve that cuts off the gas supply, the heat pump stops working and a ventilation system expels any hydrogen that may be on board.
What is the potential of hydrogen mobility by 2030?
First of all, it should be noted that hydrogen can be adapted to all types of mobility: road (bicycles, saloons, light commercial vehicles, HGVs, buses), rail (trains), sea and river (barges, cargo ships, ferries, etc.), and air (passenger planes and soon airliners), but also for special machines (forklifts, construction machinery, mining machinery, etc.). Already now, hydrogen can be used as a preferred energy source for decarbonised mobility over long distances, for HGVs or for intensive use. In fact, hydrogen offers particularly powerful solutions for all types of vehicles that require high engine power, long range or intensive use. This is the case for public transport, goods transport or household waste collection, for example. Germany has just inaugurated a fleet of fourteen 100% hydrogen trains, a world first. Developed by the French company Alstom, this technology is the preferred way to reduce CO2 emissions and replace diesel trains, especially on small regional lines where the cost of switching to electric is too high compared to the link’s profitability. Currently, half of regional trains in Europe run on diesel. Last year, France ordered 14 trains from Alstom to equip regional lines.
Long-distance transport such as trains or ships have a high need for electrical energy, which must be stored and transported to cover thousands of kilometres. Hydrogen becomes interesting when battery solutions are not suitable for the intended use or do not meet constraints such as recharging time, range or payload. There are also mixed vehicle solutions that combine an electric battery and a hydrogen fuel cell, which acts as a range extender and thus doubles the range of the electric vehicle. However, many experts believe that hydrogen does not have much of a future for passenger cars, unlike for business use. Renault and Stellantis offer hydrogen-powered light commercial vehicles in their range, but no passenger cars. In fact, hydrogen appears to be a logical choice compared to electric for commercial vehicles. There is no need to carry heavy batteries that reduce the maximum payload and the vehicle can be refuelled in minutes instead of hours. In addition, companies can set up their own hydrogen production station for their captive fleets via an electrolyser, the price of which should continue to fall. This logic also applies to buses and especially to HGVs, where hydrogen is even more popular and is being developed by brands such as Toyota, Hyundai, Daimler and Hyzon.
Like Germany, France has made hydrogen a priority area for investment with the launch in 2020 of the National Strategy for the Development of Decarbonised Hydrogen, which makes it a strategic industry with €7.2bn by 2030. State support has been reinforced by the France 2030 investment plan, which provides the sector with an additional €2 billion to support gigafactory projects for key equipment such as electrolysers, heat pumps and tanks. For its part, Europe is encouraging the sector by supporting 15 French projects with a budget of €1.5 billion. More recently, the RePower EU Plan doubles the targets for renewable hydrogen produced in Europe and imported. Hydrogen therefore appears to be an energy vector that can meet the challenges of decarbonisation and energy sovereignty.
To conclude, we can detail in a few figures the potential of hydrogen mobility described in the French Ambitions 2030 scenario, i.e. by the end of the decade:
- 1,000 to 1,700 hydrogen stations
- 300,000 to 450,000 light vehicles
- 5,000 to 10,000 HGVs
- 65 to 135 ships
- 250 trains
To which should be added an anticipated development in public transport (buses), taxi fleets and commercial vehicles. According to this trajectory and these optimistic assumptions, hydrogen would power 18% of the French vehicle fleet in 2050 and thus contribute to reducing CO2 emissions by 55 million tonnes, almost a third of the reduction required in the government’s reference scenario to achieve carbon neutrality by that time.
In conclusion, the future of the hydrogen vehicle continues to raise questions: a pipe dream for some, especially for passenger cars, essential to a decarbonised energy mix for others. The advantages it offers in terms of environmental benefits, as well as the services it provides to optimise the use of renewable energy and to give users flexibility in terms of range and charging time, justify massive public and private investments in France and Europe, with the aim of positioning France as a European leader in renewable and low-carbon hydrogen. By integrating the potential for improvement in production and use technologies, hydrogen mobility thus appears to be a technically and economically sustainable solution to the decarbonisation of transport and energy sovereignty. Even if the hydrogen car for all will not happen overnight, hydrogen appears to be a credible substitute for fossil fuels as a source of energy for long-distance mobility, as an industrial gas in heavy industries and above all as an energy vector for optimising the use of renewable energies and the functioning of the electricity and gas system. Enough to justify the recent interest in it and the optimism of the scenarios discussed in this paper concerning an energy source that has been known and studied for over two centuries.

The Hopium Machina, the first French hydrogen car, 500 hp, 250 km/h, €120,000, presented at the 2022 Paris Motor Show
----------------------------------------------------------------------------
Remember
Hydrogen vehicles: where do we stand?
Hydrogen is often seen as the fuel of the future: abundant on Earth and easy to extract, renewable and not very polluting. Despite this, the market remains small, with a very limited offering and low sales. Can hydrogen (H2) really become the fuel of a decarbonised and sustainable mobility?
Hydrogen vehicles (HVs): how do they work?
Hydrogen is rare in its natural state, but it is easy to extract and very common in combination with other substances in water and hydrocarbons. It can be used as a fuel in a combustion engine or in a fuel cell that produces electricity for use in an electric motor. Hydrogen can be produced in several ways: the most common is steam reforming of natural gas, which is simple and cheap but very polluting; charcoal can also be burnt but this process is also very polluting; the only environmentally friendly process is water electrolysis, where electricity is used to break down the water molecules and produce dihydrogen (H2) and oxygen, a simple but still very expensive process.
In recent years, many countries, including France, have adopted a Hydrogen Plan to develop production by electrolysis. The French Plan is supported by Europe.
Pros and cons of hydrogen vehicles
Hydrogen vehicles have many advantages over battery electric vehicles: the same driving comfort but a longer range and a much shorter refuelling time. But they have some shortcomings: a very underdeveloped network of stations; the very high cost of hydrogen stations (€1m per station); high price and operating costs (double that of electric vehicles). Moreover, the energy efficiency of HVs, from the electricity used at the wheel, is barely 30% and H2 is complex to store and transport. The only sustainable solution is therefore the production of H2 on the site where it is distributed.
The future of hydrogen vehicles
The future of HVs for individuals is therefore uncertain. On the other hand, its use to store intermittent electricity from renewable energies (solar, wind), on a massive scale and for a long time, has immense potential. H2 can thus give electricity and gas networks the flexibility they need. The safety of HVs in the event of a crash is a problem that has been solved by extensive crash tests on the tanks and internal leakage safety systems.
The potential of hydrogen mobility in the medium to long term is real for vehicles that require high engine power, long range or intensive use (e.g. public transport, goods transport, waste collection, etc.). Its use in trains instead of diesel is promising. It also wins when refuelling time, range or payload constraints are important. It can also be used as a range extender, doubling the range of electric vehicles. H2 is thus a logical choice compared to electric for commercial vehicles.
In conclusion, hydrogen has many advantages in terms of environmental benefits and flexibility for its users (comfort, range, refuelling time). It is a credible substitute for fossil fuels for long-distance mobility and for optimising the use of renewable energies and the electricity and gas networks.







